Hypoplastic Left Heart Syndrome
Last updated December 2021
Hypoplastic Left Heart Syndrome (HLHS) is a congenital heart condition (a problem that a baby is born with). Below is a description of the heart condition and possible treatments offered.
This information is also available in booklet format, click here to download the PDF.
On this page you will find;
-
The normal heart
-
The circulation before birth (fetal circulation)
-
The Hypoplastic Left Heart
-
Questions commonly asked at the time of diagnosis
-
Possible treatments for HLHS
-
Genetics
-
Cardiac Tests
-
First stage of treatment
-
Second stage of treatments
-
Third stage of treatment
-
The MCT Diet
-
Outcomes of life with a single ventricle heart
-
Other interventions
-
ECMO / ECLS
-
Medications
-
Challenges, consequences and complications on the single ventricle treatment pathway
-
Identifying key hospital and community staff
-
Understanding common terms
The normal heart
Understanding the normal flow of blood through the heart can be daunting so here we have described it in two ways.
The heart is a clever pump. Its job is to collect and send blood to different parts of the body. Blood contains all the things we need to make energy: oxygen, nutrients (food) and water, which it takes to every part of the body so that every part of the body has the energy it needs to work, grow and repair.
The body’s circulation has red blood that is filled with oxygen and blue blood that is empty of oxygen.
The heart has two sides. The right side’s job is to collect blue blood from the head, neck and body into the top right chamber (the right atrium). It then passes into the bottom chamber (the right ventricle) which pumps the blood to the lungs.
The lungs do their job and pass oxygen into the blood; this turns the blood red. This oxygen-filled blood then needs to be pumped around the body by the heart. The red blood is collected in the top left chamber (the left atrium) and then passes to the lower chamber (the left ventricle) that pumps the blood out of the heart and around the body.
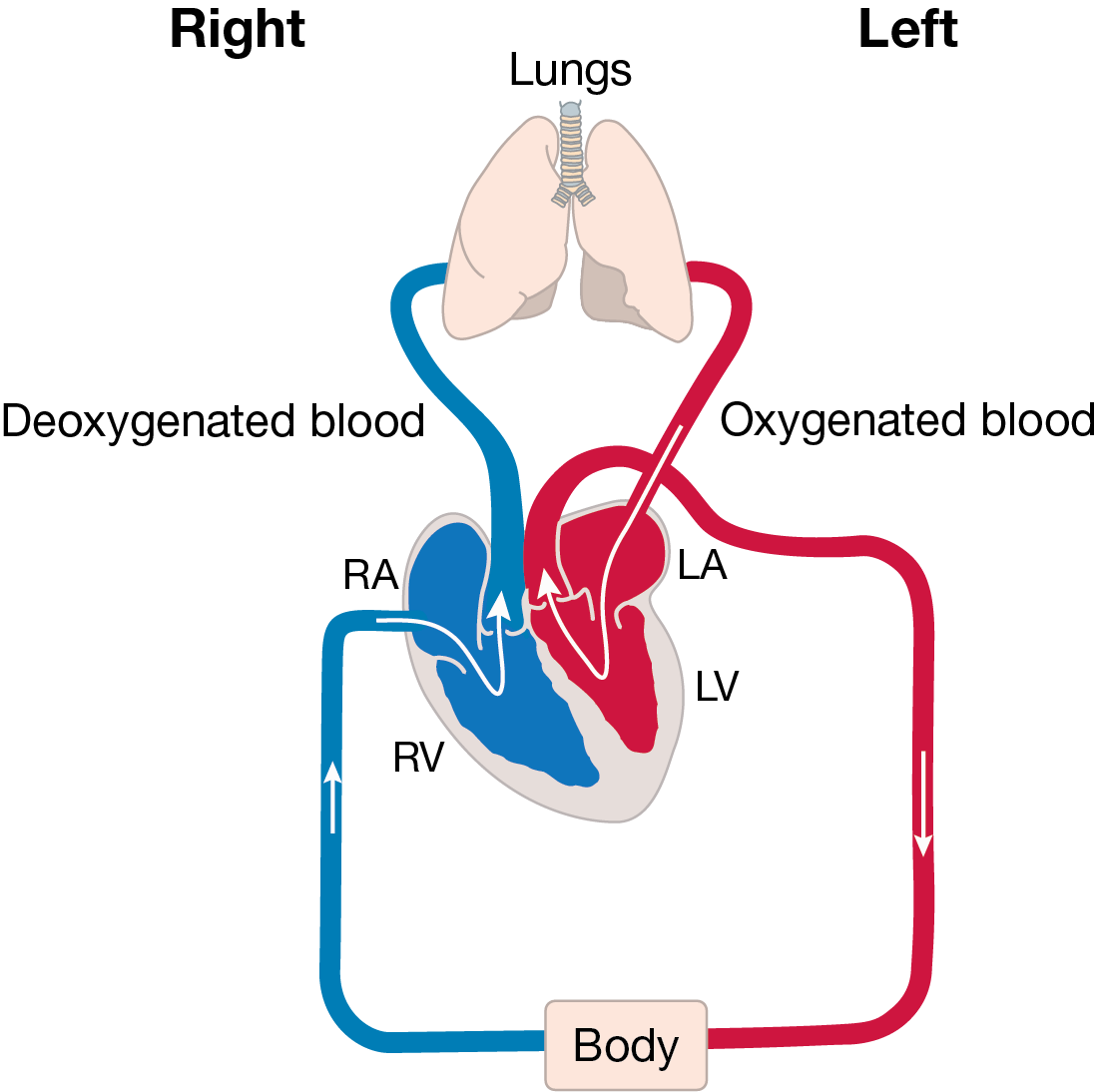
The body uses all the oxygen in the blood, turning the blood blue, and then sends it back to the right side of the heart to start the journey all over again. The following paragraphs explain the heart again but use more of the terms you may hear from your medical team and the picture looks more like a real heart.
This is how the journey begins: blood returns from the body, via the superior vena cava and the inferior vena cava, to the right side of the heart into a collecting chamber (right atrium). This blood has a bluish tinge (blue blood) because the body has taken (extracted) all the oxygen from it (deoxygenated blood).
The blood passes through a valve (tricuspid valve) into a pumping chamber (right ventricle), which then pumps the blood to the lungs via the lung arteries (pulmonary arteries).
As the blood passes through the lungs it picks up oxygen: this turns the blood a red colour (oxygenated blood). This blood flows to the left collecting chamber (left atrium) and then passes through a valve (mitral valve) to the left pumping chamber (left ventricle).
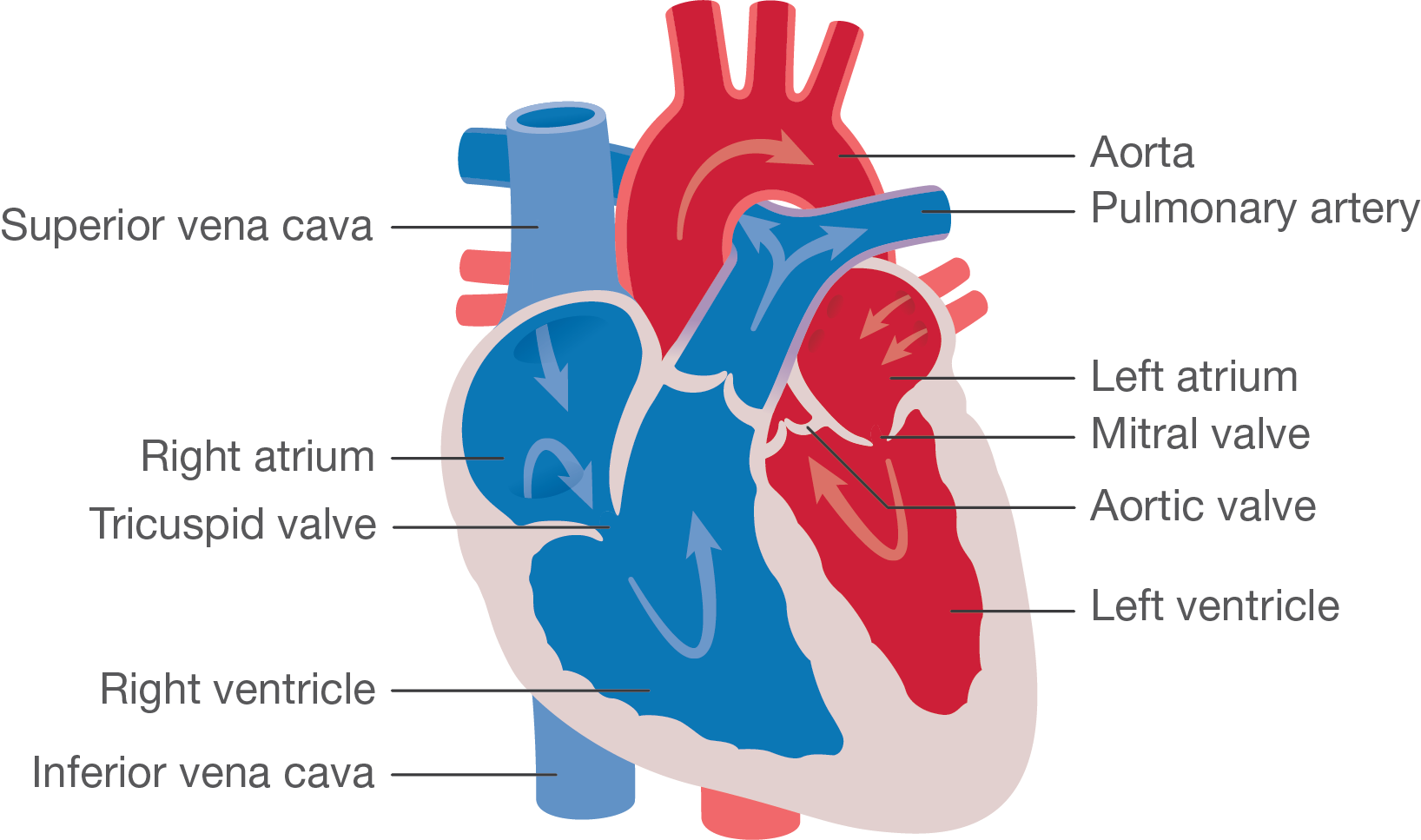
The left ventricle then pumps blood to the body through a valve (aortic valve) via the main body artery (aorta).
The body uses the oxygen from the blood to help make energy. As the oxygen is used up, the blood takes on a blue colour and needs to return to the lungs to collect more oxygen. The journey then starts again.
Below you will find an animation about the normal heart – A visual guide to help you understand the information on this page.
The circulation before birth (Fetal circulation)
When the baby is still in the mother’s womb it does not need to breathe for itself as the mother is supplying all the oxygen to the baby via the umbilical cord.
The circulation before birth is different from that after birth. It is designed so that the oxygen-filled blood from the umbilical cord goes to the most important parts of the body, for example, the brain. Very little blood needs to go to the lungs.
The heart has designed a series of bypass systems. There is a hole between the upper collecting chambers (the left and right atria) called the foramen ovale. Some oxygen-filled blood passes from the right to the left collecting chamber then on into the left pumping chamber (left ventricle) which pumps the blood around the body. Some blood continues from the right collecting chamber down into the right pumping chamber where it is pumped up to the lungs, via the pulmonary artery.
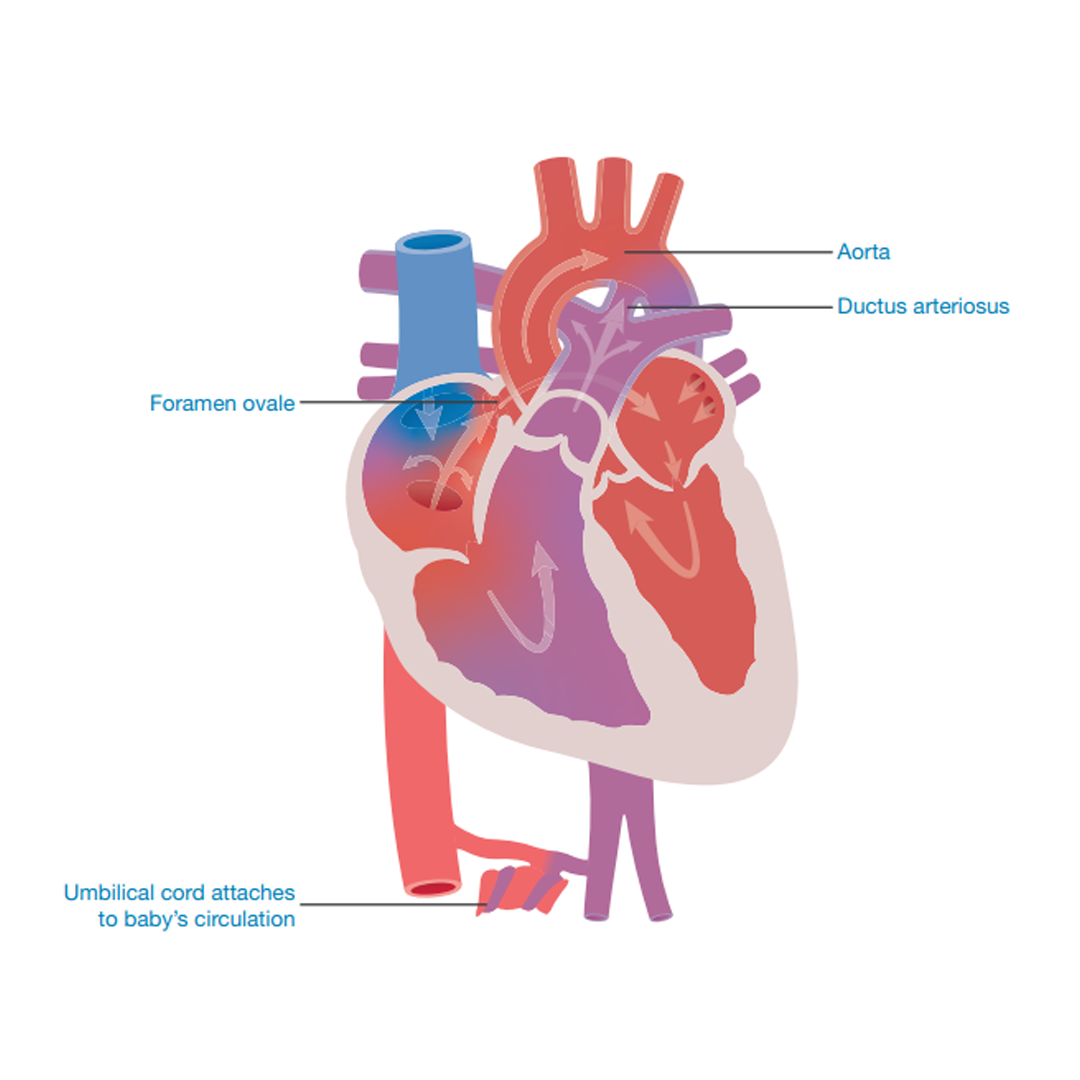
The second bypass is a connection between the lung artery (pulmonary artery) and the body artery (aorta). The connection is called the ductus arteriosus (duct). Blood passes from the right pumping chamber (right ventricle) into the lung artery (pulmonary artery). Some of the blood travels through the lungs but most of it flows through the duct to the body artery (aorta) and around the body.
When the baby is born and starts to breathe for itself, the bypass systems are no longer needed. Gradually over the first few days or weeks after birth, the duct (ductus arteriosus) and the hole (foramen ovale) between the upper two pumping chambers will close off and the baby’s circulation will be as described by the heart condition diagnosis (as outlined in the following sections).
The Hypoplastic Left Heart
Hypoplastic Left Heart Syndrome is a congenital heart condition (a condition that a baby is born with). It is made up of a collection of abnormalities on the left side of the heart. In a majority of cases, the pumping chamber (left ventricle) is small (hypoplastic) and the mitral and/or the aortic valve may be narrow, blocked or not formed at all. The body artery (aorta) is often small (hypoplastic).
In some cases there will be a hole between the two upper chambers of the heart, known as an atrial septal defect.
The blood’s journey through the heart is very different from normal. The blue (deoxygenated) blood flows into the right collecting chamber (right atrium), through the valve (tricuspid valve) into the right pumping chamber (right ventricle). From there it is pumped to the lungs where the blood receives oxygen. This is like the normal heart.
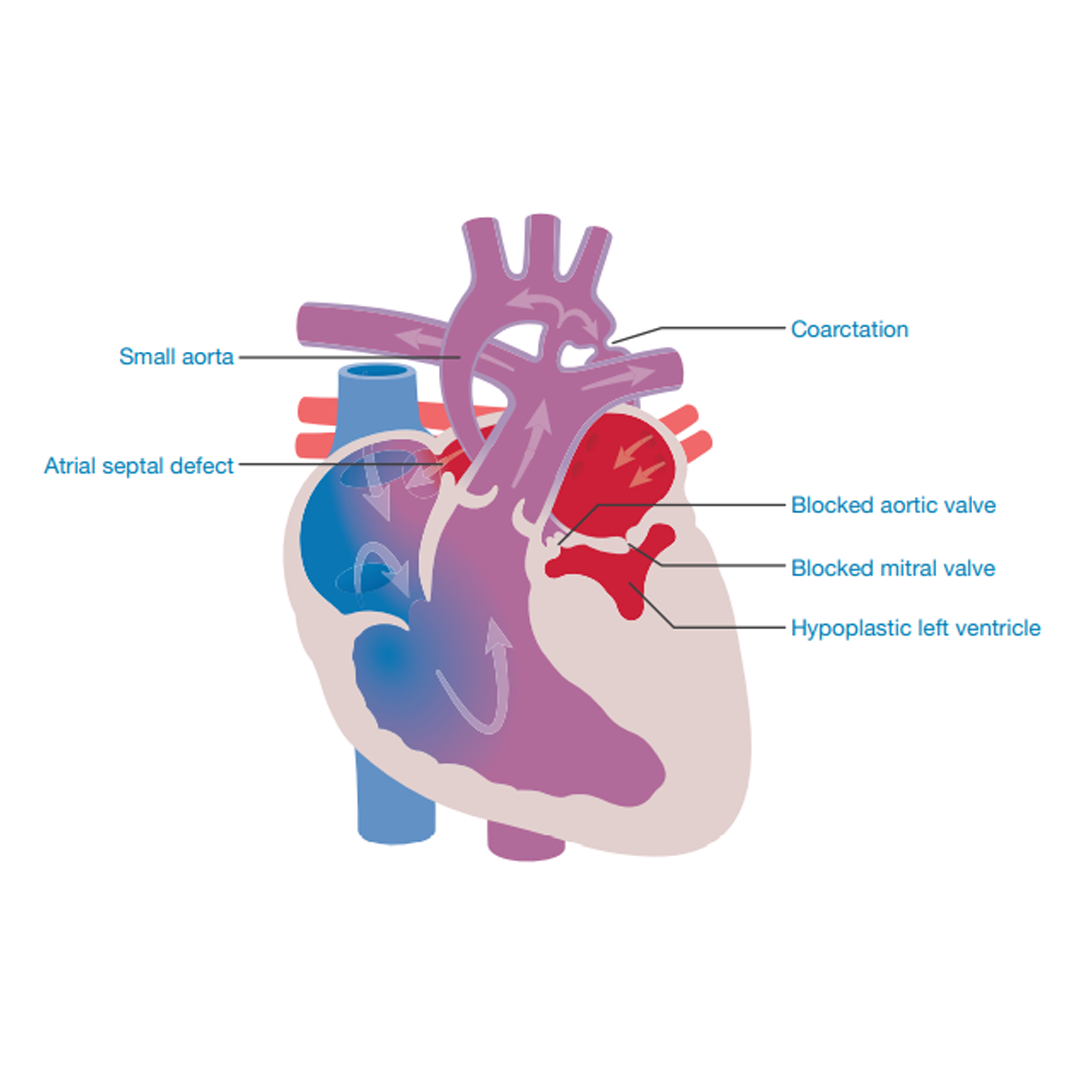
The red oxygen-filled blood then flows from the lungs into the collecting chamber (left atrium) in the left side of the heart, but it will be unable to then pass into the left pumping chamber (left ventricle), as the mitral valve between the two chambers will be small or blocked.
The blood has no choice but to return to the left collecting chamber from where it then passes through the hole (atrial septal defect / foramen ovale) between the two collecting chambers into the right side of the heart, where it mixes with the blue blood and follows the path through the lung valve (pulmonary valve) to the lungs.
This structure does not allow for oxygen-filled blood to move into the body. The only way for oxygen-filled blood to reach the body is through the ductus arteriosus, the duct between the lung artery (pulmonary artery) and the main body artery (aorta). As the mixture of the blue and red blood passes through the lung artery, some of the blood passes down the open (patent) ductus tube into the aorta and then onwards to the baby’s body. The mixture of blood does mean that the baby is blue (cyanosed) as the blood is not totally filled with oxygen but it also means that some oxygen is available to make energy.
Both the foramen ovale and the ductus arteriosus normally close after birth. It is at this point that many babies with a heart condition start to have difficulty getting blood and oxygen around their body. Medical teams will start an infusion of Prostaglandin, a drug that mimics the mother’s hormones, to slow the closure of the ductus arteriosus. This allows the baby time to recover from birth and the medical team time to plan and discuss the first treatments.
Below you will find an animation about HLHS – A visual guide to help you understand the information on this page.
Incidence
Hypoplastic Left Heart Syndrome is a fairly rare problem that occurs in approximately 1 in 4,000 babies and accounts for 1% of all congenital heart disorders.
Questions commonly asked at the time of diagnosis
What causes the heart to form this way?
As yet the causes of single ventricle heart conditions are unknown. They cannot be linked with mild drinking or the taking of ‘over the counter’ or most prescribed medications.
There are some indications that these conditions can be linked to heavy smoking, alcohol or drug abuse. Anyone with diabetes has an increased risk of having a child with a heart condition.
It is very likely that one or more abnormalities are present before the third month of pregnancy but they may become more obvious as the heart is growing through the second half of the pregnancy.
Heart abnormalities can be present in babies who have other congenital problems, for example, genetic disorders like Edwards’ syndrome or DiGeorge syndrome (22q Deletion).
Why was the problem not detected during pregnancy?
It is possible to detect single ventricle heart disease during pregnancy but it may not be obvious during scans due to the size or position of the baby. In some cases the condition can become more evident later in pregnancy after the routine scans have been performed.
Why is the condition treatable but not correctable?
Doctors have developed a way of bypassing the malformations within the heart so that blood can circulate around the body but they are unable to make a ventricle, so even following surgery the heart will only have one working pump rather than the two seen in a normal heart.
What does palliative mean?
Palliative treatment means that the treatment is unable to cure the problem. In the case of the treatment for a complex heart problem it means that the treatment can bypass but not cure or totally correct the problem.
Can my baby have treatment anywhere in the country?
Most – but not all – congenital heart centres around the country offer surgery for these conditions. It may be necessary for your baby to move hospital to receive treatment. This would be discussed with you at the time of diagnosis.
Genetics
Many families who have a child with congenital heart disease ask why their child was born with the condition. In some cases the malformation will have occurred because of a genetic problem that has affected the heart as it formed in the womb.
A blueprint
When a new house is built, the architect draws up a plan of what the house will look like and how it is going to be built. When a new child is being created, information is drawn from the mother and the father to make a plan of how the child will look and how their body will be put together to work.
A genetic blueprint
Every person in the world has a genetic blueprint of their own. The blueprint is stored in every cell in their body and holds the information required to help the body grow, develop and work properly. The information is made up of lots of messages which we call genes. We have about 30,000 genes in each cell of our body. Different genes carry different messages that are responsible for instructing our body to do specific things. For instance, genes determine the colour of our hair or eyes or how cells work in different organs, for example, the liver, heart or lungs. The genetic blueprint is our very own information computer.
The genes are stored in coils and split into chromosomes (see adjacent diagram). Humans have 23 pairs of chromosomes, so 46 in total. 23 chromosomes (one of each pair) come from the mother and the other 23 from the father. They are transferred in the egg and the sperm that make a child.
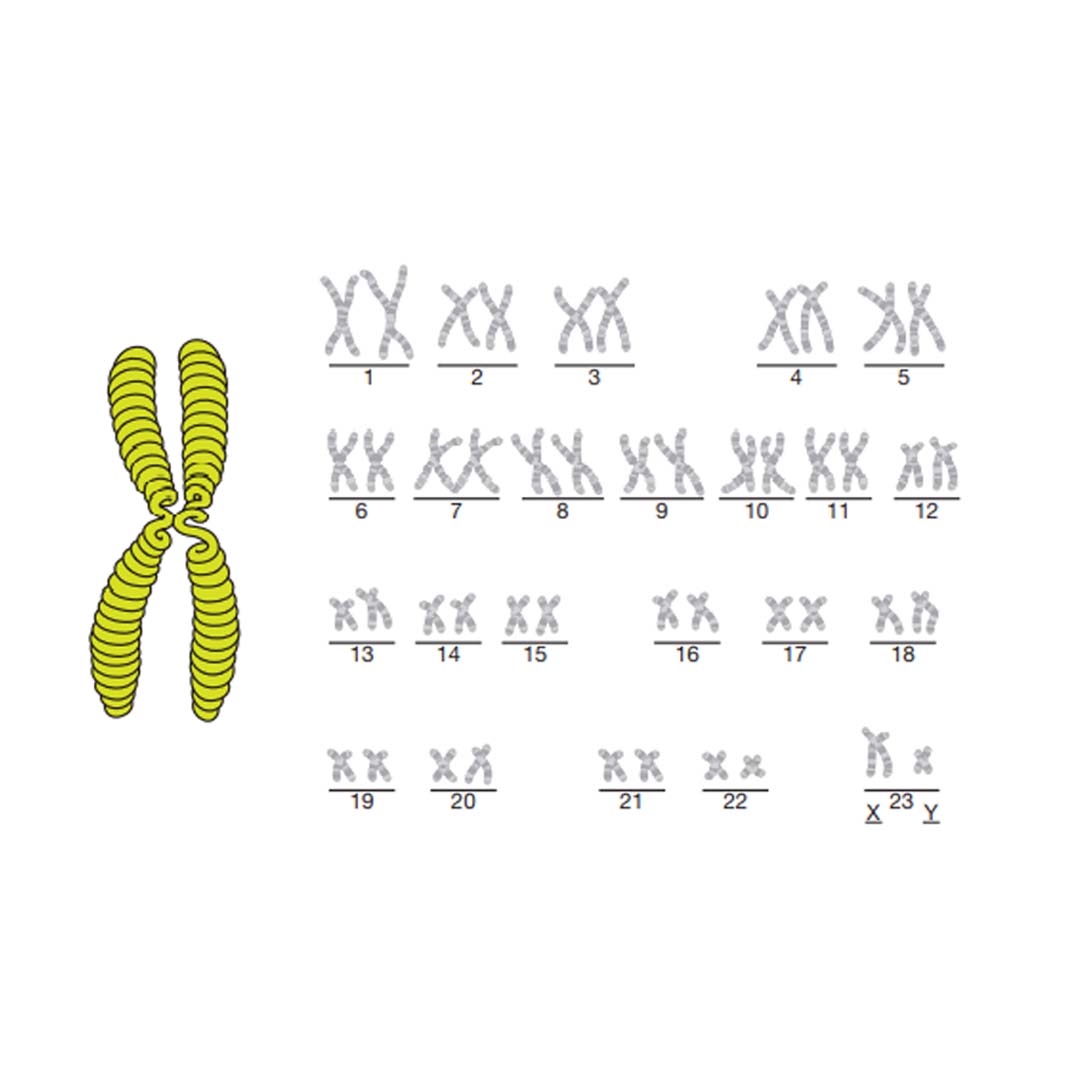
The picture above shows what the chromosomes look like – if you look at the last set of chromosomes in this picture you will see that they are labelled X and Y. These are the chromosomes that decide if you are male or female. The chromosomes picture must represent a man because there is an X and a Y chromosome. A woman always has two X chromosomes. The sex of a baby is determined by which chromosome comes from the father: X for a girl, Y for a boy.
Genetics and heart disease
Some congenital heart conditions are linked to a genetic disorder such as Edwards’ syndrome or 22q Deletion. These disorders are
caused when one of the 46 information chromosomes is malformed. As the baby grows in the womb, the genetic malformation will cause a particular part of the heart to develop incorrectly. In some cases the genetic condition can be detected before birth.
Geneticists (genetics doctors) are always looking for genetic causes for congenital conditions; however, there are many heart conditions that do not have a specific genetic cause. Many of the single ventricle heart conditions fall into this group.
There may be many factors, as yet unknown, that cause a baby’s heart to develop abnormally. The problems can occur early in pregnancy as the heart forms (before three months) or later as the heart grows.
Congenital heart conditions occur in 1 in 130 pregnancies. If you have previously had a child with a single ventricle heart condition the risk of it happening again rises to between 5 and 8%. There is also a risk to any pregnancies in the person with the condition and their brothers and sisters. Although the risk is higher, over 90% of future babies will have no problems with their heart.
Possible treatments for Hypoplastic Left Heart Syndrome
Having explained your child’s diagnosis, the doctor will go on to discuss what treatments are available. Although none of the treatments provide a long-term cure, for many children they can offer a reasonable quality of life.
It is vital that parents are fully included in the treatment discussions and decisions taken for their child. Parents are their child’s advocates until they reach an age when they can be involved in discussions about their own treatment. These treatment options may include:
Surgical treatment
The aim of surgical treatment is to re-route the blood through the right side of the heart, bypassing the blockages on the left side. This surgery is offered in three stages over three or more years and involves extremely high-risk, open-heart surgery. Your doctors will explain the individual risk for your child.
Even after all the surgery has been performed, your child’s heart will not look normal or work normally. Most children have a good quality of life although they will always have less energy than their peers. Eventually the heart will tire and may fail, if this is the case they may be considered for heart transplant. More information on the surgery is provided below.
Supported comfort care
As the long-term outcome is unclear for children with Hypoplastic Left Heart Syndrome, it is important to know that there is the option to not put a child forward for surgery. This would be discussed fully with you by your medical team. The baby would be allowed to die peacefully either in the hospital, at a local children’s hospice or at home; the family would be fully supported by hospital and community nursing and medical staff.
Transplantation
Heart transplantation is one of the possible treatments for single ventricle heart disease, but it is not offered as a first treatment within the United Kingdom for the following reasons;
• Very few donor hearts small enough for a baby are available in the United Kingdom and babies with a single ventricle heart would not survive long enough for a donor to become available.
• Transplanted hearts do not last for ever and there are many risks involved throughout the recipient’s life. Offering surgery as a first treatment path and retaining transplant as a future option offers a greater chance of a long life for a child
Cardiac Tests
It is important that the cardiac team have as much information about the baby’s heart or circulation as possible. A number of tests are available to help them as they plan treatment and assess recovery.
Oxygen saturation
This is a simple painless test where a plastic strip or clip is attached to a finger or foot to measure the amount of oxygen circulating in the blood of babies, children and adults. It is particularly important for children with complex heart conditions who are blue (cyanosed) because of low oxygen levels as it helps the medical team to plan their treatment and care.
Blood tests
Blood is a very important part of the circulation because it carries all sorts of things around the body: oxygen, nutrients and chemicals. Taking samples of blood can be a very helpful way of monitoring how well a baby, child or adult is and how the body is coping with an illness or long-term condition.
Chest X-rays
For doctors to see if a baby’s, child’s or adult’s chest is healthy, especially before and after surgery, the easiest test is to take an X-ray of the chest, so that they can see how the chest is healing but also to look for infections or fluid collections in the lung area. These tests help plan treatment and post-surgery recovery support.
Echocardiogram (Echo)
Echocardiography uses sound waves to scan the heart which is then shown as a picture. This is the same type of scan that mothers have during pregnancy. The test does not hurt, but children sometimes have a light sleeping medicine (sedative) so that they stay still. This is the first test that will be done on the baby, as it is the easiest and least stressful way of making a diagnosis of congenital heart disease.
ECG
Electrocardiography (ECG) is the recording of the electrical activity of the heart. The heart has its own electrical system, which passes the message to beat (to pump) through the heart’s muscle tissue. Sometimes the message breaks down and so by recording the rhythm, the medical team can see if the messages are being passed properly. It is also possible to assess the size of the pumping chamber and the thickness of the heart wall.
Cardiac catheterisation
It is often necessary for the doctors to gain more information about the heart than an Echo or ECG can provide, so the child may need to undergo cardiac catheterisation. This investigation is best done with the child asleep, so they will be given a light general anaesthetic. Teenagers and adults may be awake during the tests but they will be given an injection to anaesthetise the place where the catheter tube/wires are inserted. A long thin tube (catheter) is passed into the heart via a big blood vessel, either from the groin or the neck. Through this tube a dye (radio-opaque fluid) is injected that can be seen on X-ray. The dye passes into the heart and X-ray pictures can be recorded. The doctor is then able to see the structure of the heart more clearly. It is also possible to measure the blood pressure in the different chambers and blood vessels. This information is extremely important when deciding what further treatments can be offered safely. This test is often performed whilst planning the timing and type of the next stage of surgery.
MRI scan
MRI (Magnetic Resonance Imaging) scans use radio-frequency radiation to gain a picture of the soft tissues of the heart from different angles. This can be very useful when looking at the results of surgery. Children need to lie still for this scan, meaning that many of them need some sedation or a light general anaesthetic.
CT scan
Computerised tomography, also known as a CT or CAT scan, uses X-rays and a computer to create detailed images of the inside of the body. The pictures are taken in small slices and then pieced together to create a multi-view of different sections of the body.
First stage of treatment – Norwood Procedure (Stage one)
This operation is normally performed within a few days of birth in the case of a diagnosis of Hypoplastic Left Heart Syndrome while
the ductus arteriosus (connection which joins the lung artery and the body artery) is being kept open with Prostaglandin therapy.
The aims of the first operation are:
• To improve the flow of red blood (oxygenated blood) around the body by attaching the base (trunk) of the lung artery (pulmonary artery) to the body artery (aorta). It may be necessary to enlarge the body artery with a patch.
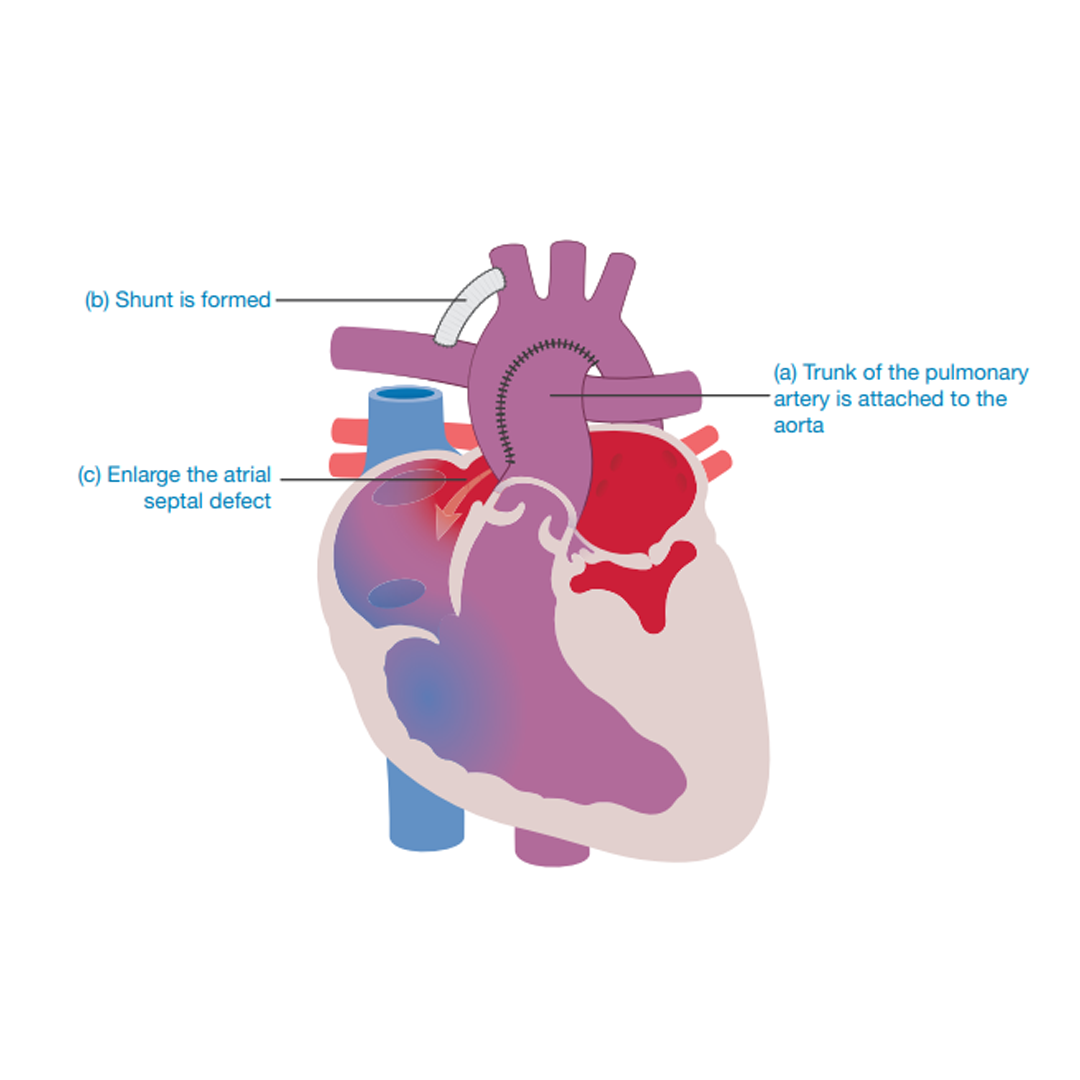
• To provide a blood flow to the lungs through a tube (shunt), creating a link between the lung artery (pulmonary artery) and the body artery (aorta). The tube is made out of soft plastic (Gore-Tex).
• To create a permanent passage (septectomy) between the collecting chambers (left and right atria) ensuring that a mix of red (oxygenated) blood and blue (deoxygenated) blood is flowing around the body.
This operation is extremely complicated and holds a high risk (chance of the baby dying). Ask your surgeon about the risk for your child. The baby may need to be in hospital for some weeks following this operation to ensure that their condition is stable before they are allowed to go home.
Modified Norwood Procedure(Norwood with a Sano Shunt)
The aims of this operation are the same as described on the previous pages for the more classic Norwood but the shunt taking
blood to the lungs is different.
The aims are:
• To improve the flow of red (oxygenated) blood around the body by attaching the base (trunk) of the lung artery (pulmonary artery) to the body artery (aorta). It may be necessary to enlarge the body artery with a patch.
• To provide a blood flow to the lungs via a Gore-Tex tube that leads from the right pumping chamber (right ventricle) to the left lung vessel (pulmonary artery). This connection ensures that blood reaches the lungs where it collects oxygen that then flows around the body
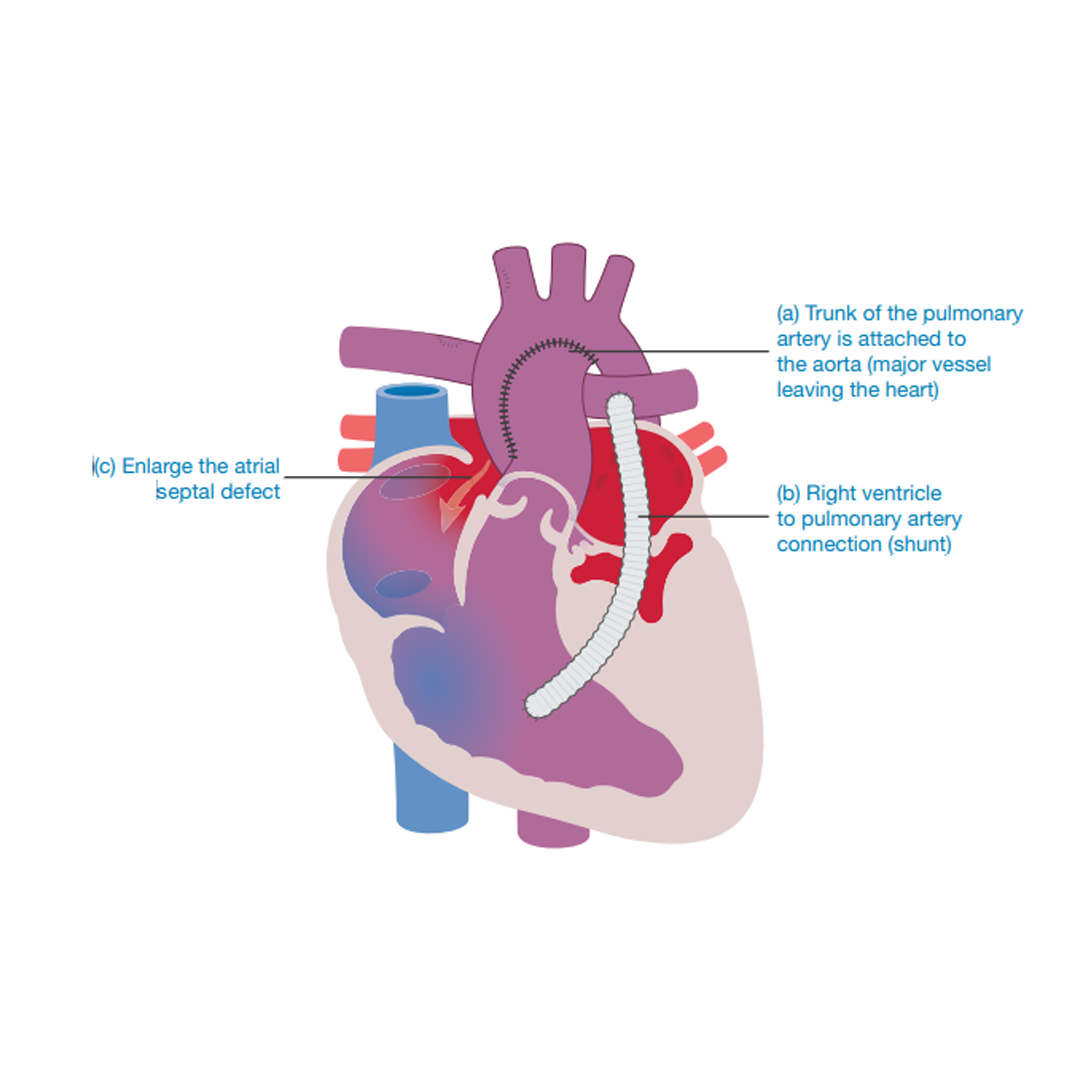
To create a permanent passage (septectomy) between the collecting chambers (left and right atria) ensuring that a mix of red (oxygenated) blood and blue (deoxygenated) blood is flowing around the body.
This, like the previously described procedure, is an extremely complicated operation and carries a high risk (chance of the baby dying). Ask your surgeon about the risks for your child.
The baby may need to be in hospital for some weeks following this operation to ensure their condition is stable before they are allowed to go home.
The Hybrid Procedure
In the past many new-born babies were not suitable for the Norwood Procedure for Hypoplastic Left Heart Syndrome because they were too small or too sick and the risks were too high.
Medical teams developed a procedure that will support a new-born baby’s heart until he or she is big enough to undergo the more complex, reconstructive Norwood operation described earlier. This procedure is called the Hybrid Procedure.
The aim of the Hybrid Procedure is to allow a flow of oxygen-filled red blood (oxygenated blood) to reach the body and to allow used blue blood (deoxygenated blood) to reach the lungs where it can collect oxygen again.
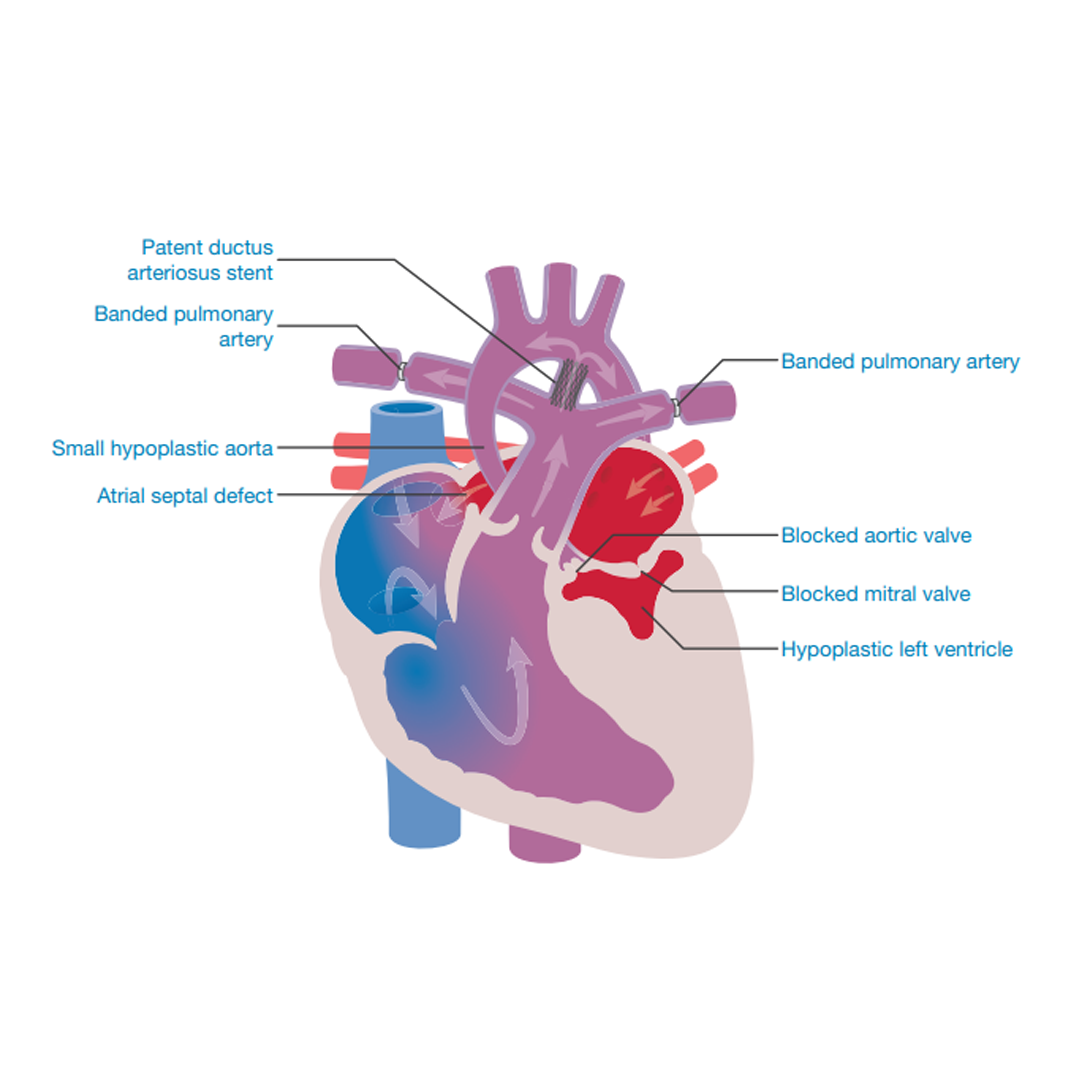
This is done by keeping open the arterial duct (patent ductus arteriosus) longer than Prostaglandins will allow. The hole between the upper chambers (the patent foramen ovale/atrial septal defect) is opened up if required. At the same time the lung blood vessels (pulmonary arteries) are protected so that they can be used during later surgery, see below.
The Hybrid Procedure is performed under a general anaesthetic by both surgeons and cardiologists working together.
There are three parts to this procedure:
1. Small bands are tightened around the left and right lung arteries (pulmonary arteries) to control the flow of blood and pressure in the lungs. This protects the pressure in the lungs for the final Fontan procedure.
2. A mesh of fine metal (stent) is placed in the patent ductus arteriosus (between the pulmonary arteries and the aorta) to keep it open.
3. At the same time the hole between the upper collecting chambers (atria) may be made bigger and permanent (atrial septostomy). This may be delayed for about a week to assess the flow of blood created by the other two parts of the procedure.
In very small and sick babies, this procedure may offer a lower risk than the full Norwood operation but the risks of this operation are still high because the baby is very small.
Children who have undergone the Hybrid Procedure will need a more complex operation at Stage two as it will combine both the reconstructive surgery of the usual Stage one Norwood Procedure and the Stage two Cavo-Pulmonary Connection.
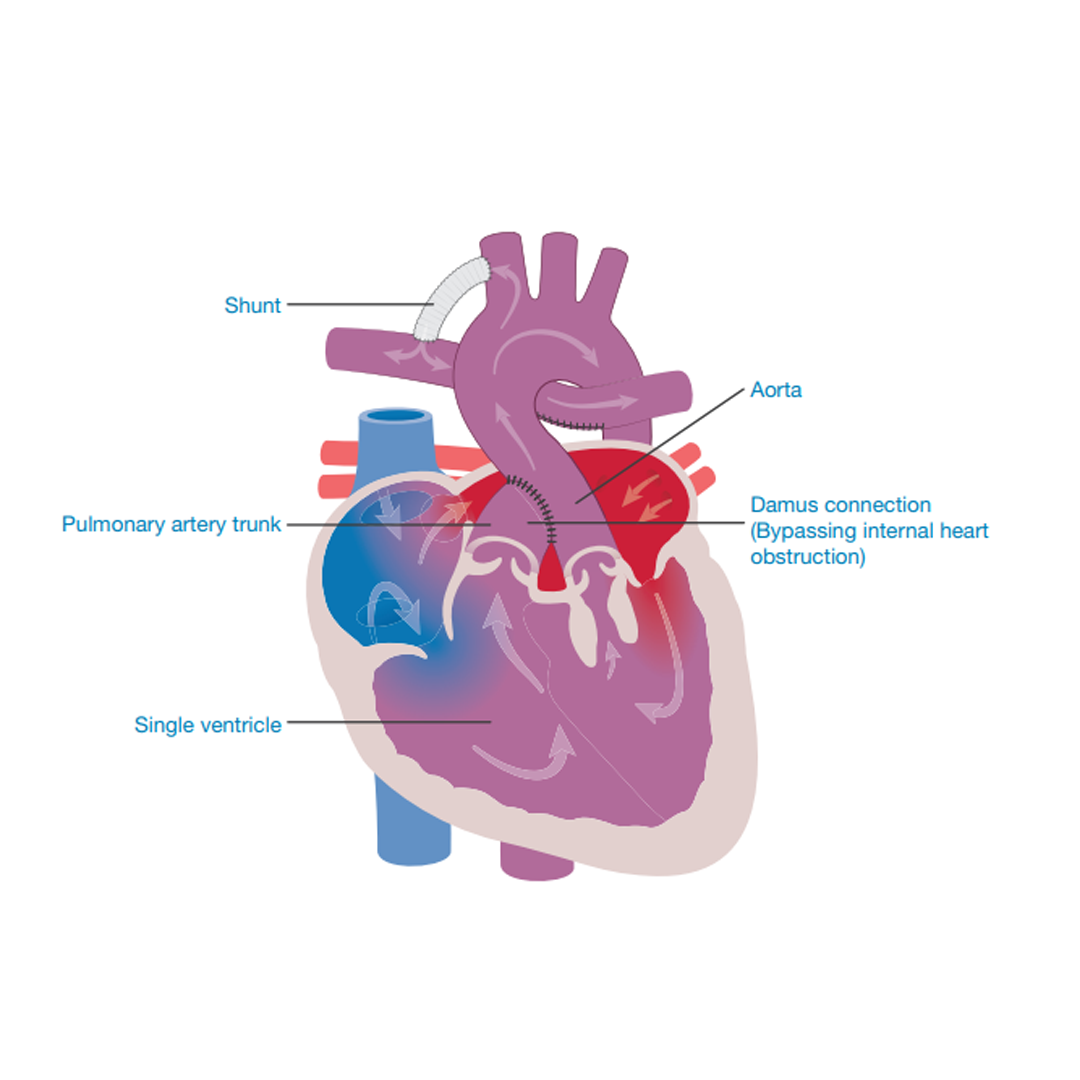
The Damus Procedure
Children who are born with only one of the two chambers (ventricles) needed to pump blood around their lungs or body and who may also have a narrowing of the body artery (aorta) may need to have surgery to aid circulation of blood from the heart to the body.
The base of the lung artery (pulmonary artery trunk) is attached to the base of the body artery (aorta). This ensures that oxygen-filled blood from the one working heart pump is directed around the body.
To ensure that blood also reaches the lungs, a connection (shunt) is made between the body circulation (aorta) and the lung artery (pulmonary artery).
Second stage of treatment – Cavo-Pulmonary Connection
Bi-directional Cavo-Pulmonary Shunt (Glenn Shunt) or the Hemi-Fontan operation
As the children grow, they will require a more permanent supply of blood to their lungs than earlier surgical procedures can provide. A Cavo-Pulmonary Connection is normally performed at between three and twelve months of age.
The aim of this operation is to take the first step to creating a greater flow of returning blue blood (deoxygenated blood) to the
lungs by redirecting the flow of blue blood (deoxygenated blood) to the lungs by attaching the upper body vein (superior vena cava)
directly on to the lung artery (pulmonary artery). The Gore-Tex shunt or the band that was inserted at the previous operation will be taken away.
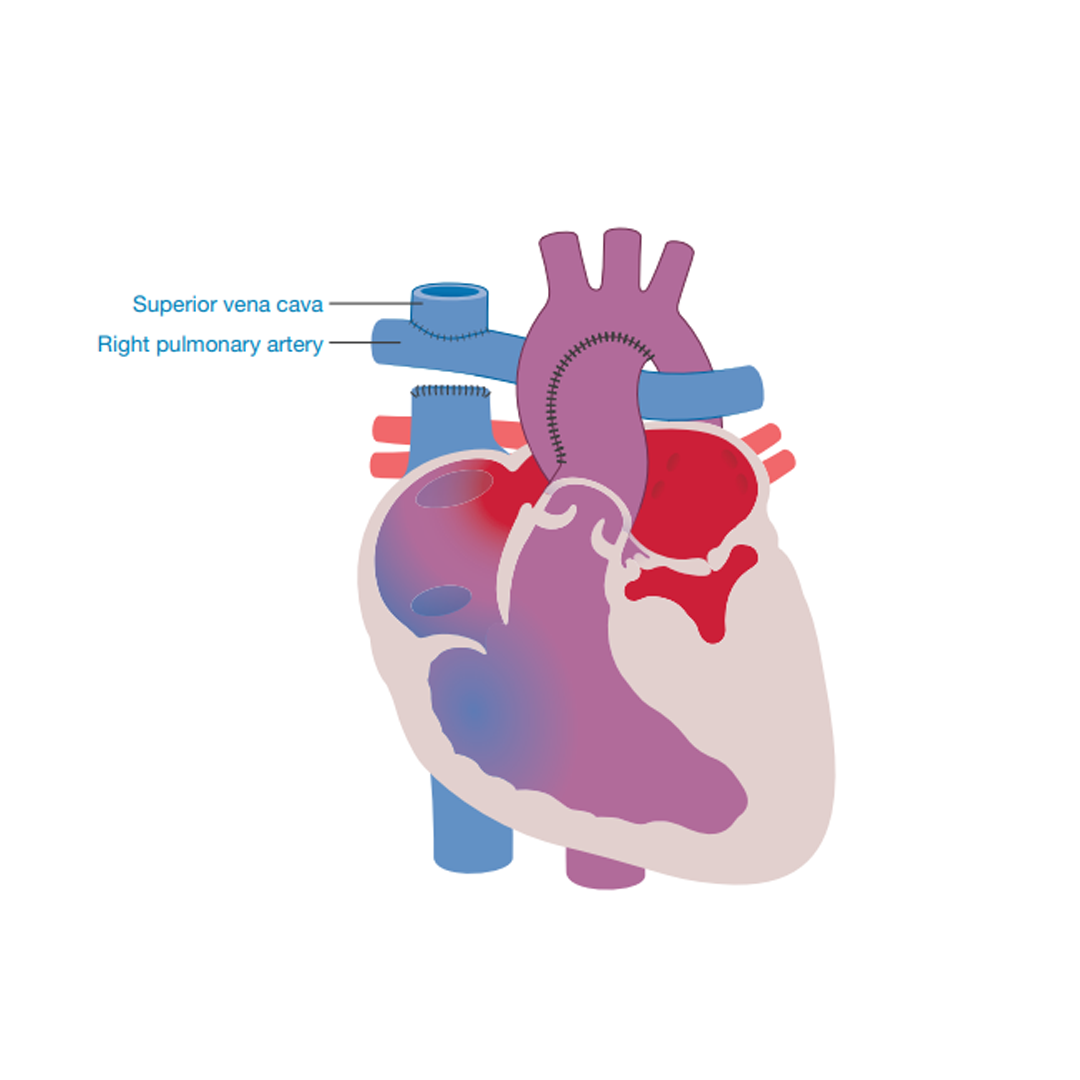
In some hospitals the stump of the lower body vein (inferior vena cava) is attached to the base of the lung artery (pulmonary artery). This is known as the Hemi-Fontan operation. Although blood does not flow through this attachment at this time, it prepares the child for the completion of a Fontan procedure at Stage three.
In most cases these operations have fewer risks than the first stage but it is important to talk with the medical team to find out what the individual risks are for your child.
The recovery time for this operation is normally short, with many children only staying in hospital for less than a week.
Third stage of treatment The Fontan Procedure or Total Cavo-Pulmonary Connection (TCPC)
The final stage of surgery will be performed as the child starts to show that they need more blood flow to the lungs. They may become more breathless on exercise or their growth may slow down. For some children this will occur before they start school. Others may be able to wait a little longer.
For more information, click here to read the LHM booklet The Fontan procedure.
The Fontan procedure or Total Cavo-Pulmonary Connection aims to:
More permanently separate the blue (deoxygenated) blood supply and the red (oxygenated) blood supply.
Increase the amount of blue (deoxygenated) blood flow to the lungs, improving the amount of oxygen flowing around the body.
Although this does not make the heart function normal, it does allow the children to grow and enjoy more physical activity. The procedure can be performed in two ways.
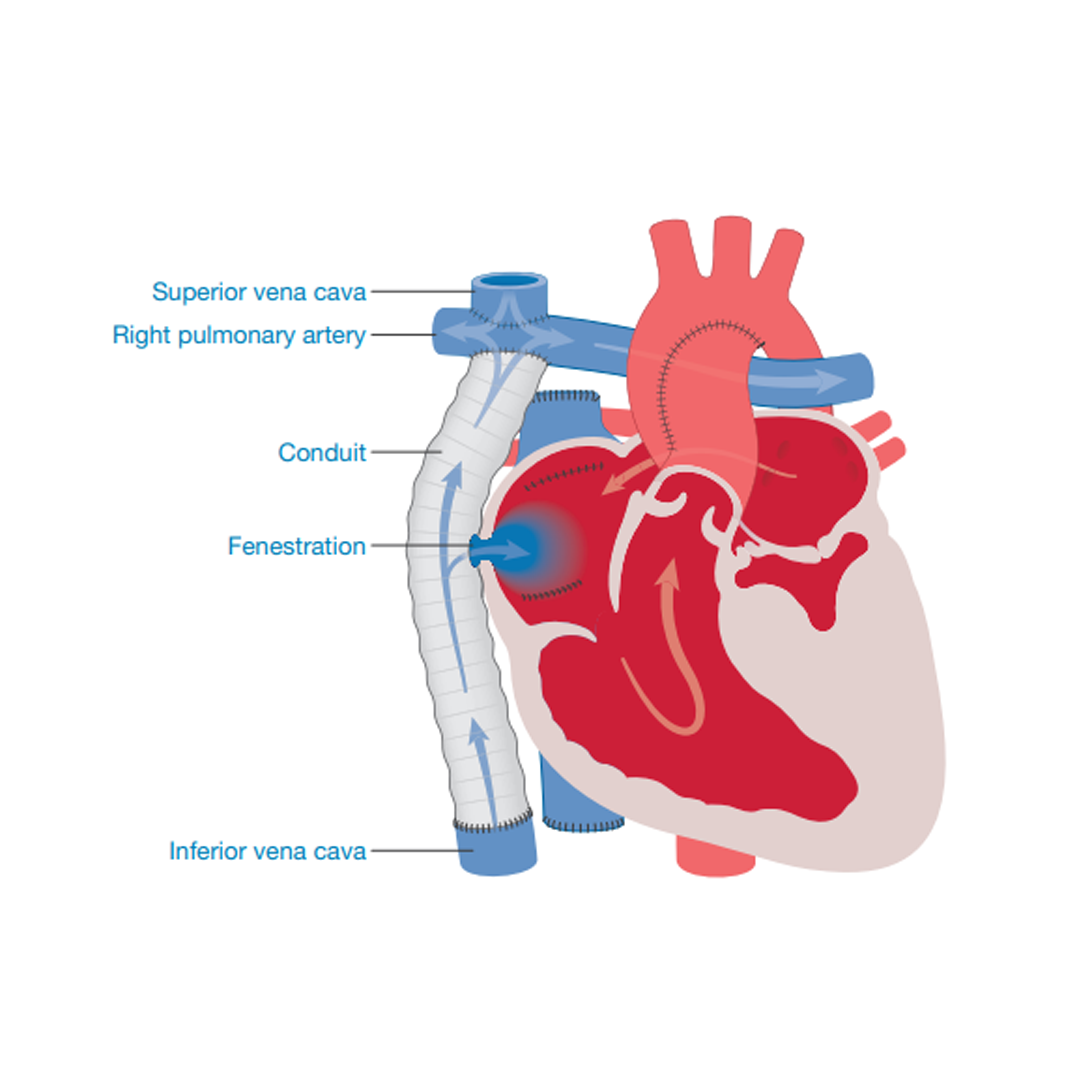
External Fontan
The external Fontan/Cavo-Pulmonary Connection is created by attaching a tube of a special plastic (a conduit of Gore-Tex) from the lower body vein (inferior vena cava) to the base of the lung artery (pulmonary artery), diverting blue (deoxygenated) blood away from the heart straight to the lungs. After this procedure all blue blood returning from the head and neck and the body will flow straight to the lungs; there is no longer a pump to drive it onwards.
The change in circulation can create a rise in the blood pressure within the lung arteries. To help reduce the pressure and any backflow, a hole (fenestration) may be created between the tube (conduit) and the right collecting chamber (right atrium). The hole acts as a protective pressure valve.
Although the initial recovery from surgery is usually swift (children are often in intensive care for less than a day), children may need to be in hospital for some weeks after the Fontan operation as it is important to give them time to adjust to their new circulation.
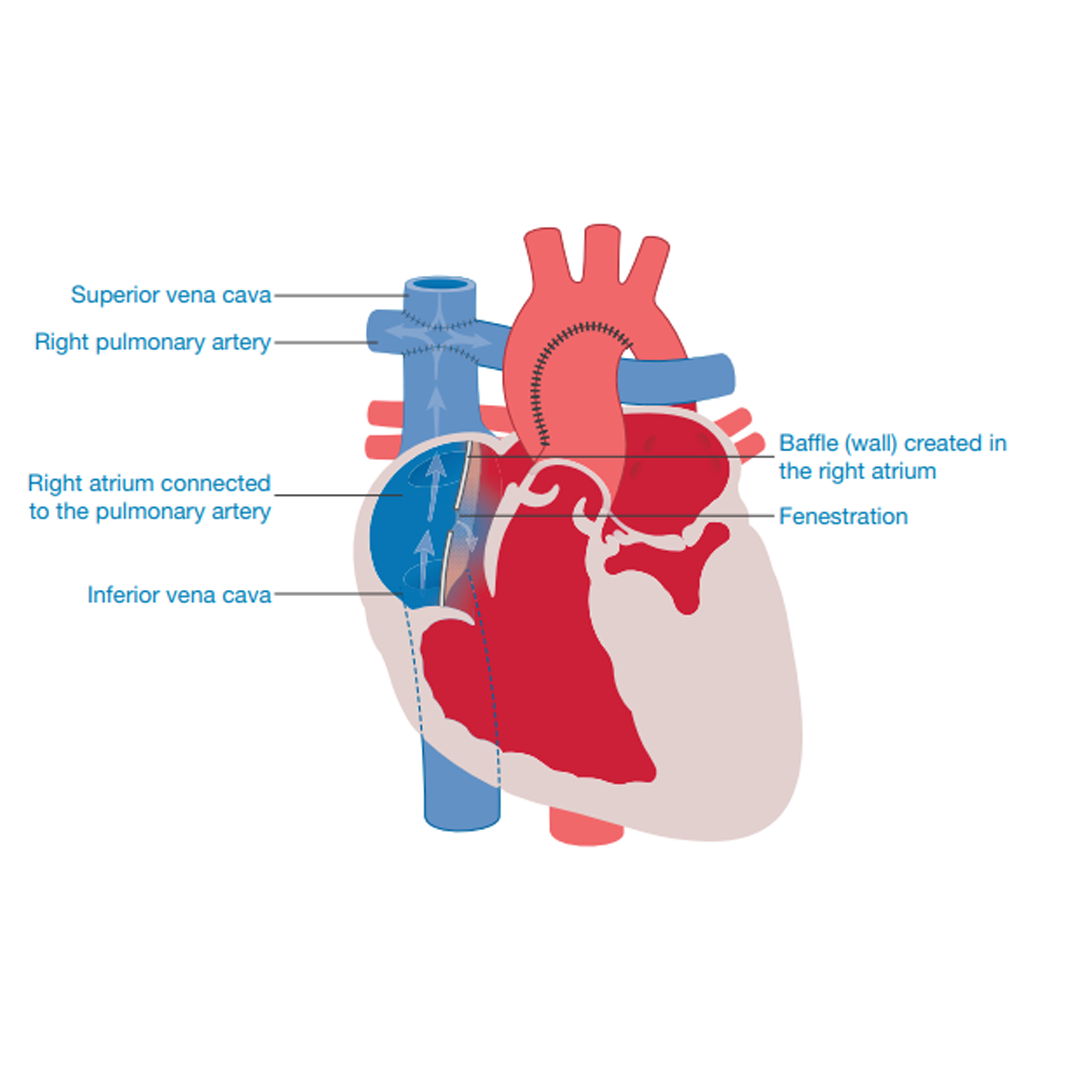
Internal Fontan
The internal Fontan is formed by creating a wall (baffle) in the right collecting chamber (right atrium) and then attaching the chamber
to the base of the lung artery (pulmonary artery). This may have been partially completed at Stage two, the Hemi-Fontan.
All the returning blue (deoxygenated) blood will now be flowing to the lungs, without a pump behind it. This causes an increase in pressure within the lung blood vessels so, to aid circulation, a small hole (fenestration) may be created in the wall (baffle). This acts as a pressure release valve whilst the child’s body adjusts to the new circulation.
Although the initial recovery from surgery is usually swift (children are often in intensive care for less than a day), children may need to be in hospital for some weeks after the Fontan operation as it is important to give them time to adjust to their new circulation.
The MCT Diet
The MCT (Medium Chain Triglycerides) diet is a special diet which is sometimes required by children for a short period of time in hospital after heart or chest surgery or for a short period of time at home.
It allows a special type of fat which is not absorbed into the lymph system. If your child requires this diet, the medical staff at your child’s hospital will advise you of the requirement, and the dietitian will help you to understand how to follow the diet at home, and when the diet is finished how to go back to your child’s usual diet. The dietitian will also provide recipes and food supplements as required.
Outcomes and life with a single ventricle heart
Survival following surgery for Hypoplastic Left Heart Syndrome (HLHS)
Understanding how well your baby is likely to do after each stage of surgery needed for HLHS can be frightening but it can also help you to understand the risks along the pathway of treatment offered to your child. It is important to remember that each baby will have individual factors which will change his or her chances of surviving each stage. Talk to your medical team about the risks they would give to your child.
It is important to remember that some babies diagnosed before birth may die either in the womb or before surgery can be offered. We do not know exactly, but we think this can happen in up to eight out of 100 pregnancies (8%). There are some babies who will have a condition
that is so complex at birth they cannot be offered any treatment.
Below we set out the results for children with HLHS who could be offered surgery.
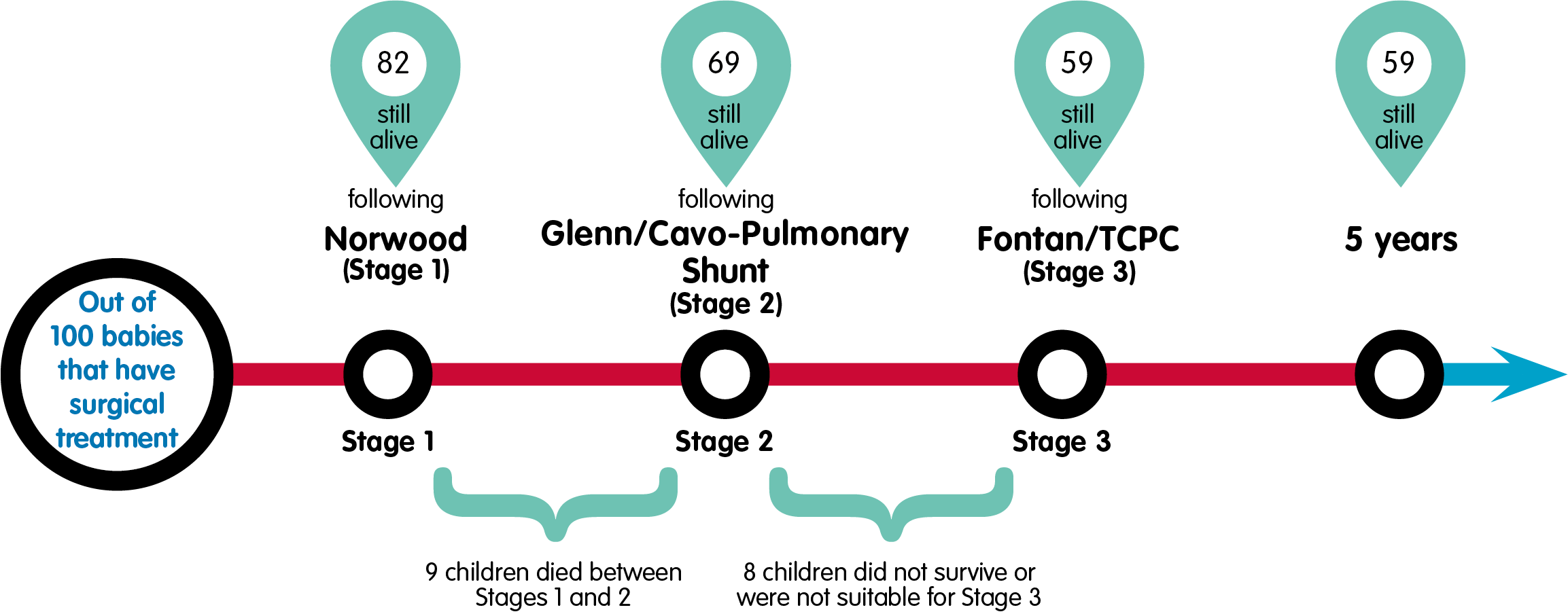
The following information shows us the outcome for the 742 babies who underwent surgery for HLHS in England and Wales between 2006 and 2017, with the information about their survival included until 2020.
These are babies who underwent either a Norwood with a BT shunt or a Norwood with Sano modification.
We have translated the numbers to show what happens to 100 babies starting down the treatment pathway; each number is a percentage of those undergoing their first operation.
The Norwood Procedure
Out of 100 babies who underwent the Norwood operation, 82 survived the operation and their hospital stay. A further 9 babies died before they reached their second operation (sometimes called interstage mortality).
The Glenn or Cavo-Pulmonary Connection
Of the original 100 babies, 73 went on to have their Stage two operation, the Glenn/Cavo-Pulmonary Shunt. 69 of those babies survived to go home. This means four babies did not survive to go home after Stage two.
The Fontan or Total Cavo-Pulmonary Connection
Not all of the children who had the second stage of surgery were suitable for the third stage of surgery. This may be because their
lung pressures were high or the flow of blood through the heart was further restricted. They may have been offered different treatment but they could not have the Fontan operation and so did not follow the Norwood pathway. Of the original 100 babies who started the Norwood pathway approximately 61 went on to have their Stage three Fontan/Total Cavo-Pulmonary Connection operation and 59 went home. Another way to look at this information is to say that out of the original 100 children who completed the Norwood treatment pathway, 59 were still alive at the age of five. It is important to remember that not all children have their Fontan operation before the age of five, so their longer-term survival may not be included in this number. Out of the 100 children, one received a heart transplant at some point on the pathway.
The Hybrid
Some babies start their surgical journey with the Hybrid Procedure. The alternative treatment pathway called the Hybrid, is offered to a much smaller number of babies than the Norwood pathway, with around one in ten babies undergoing this in the last decade in England and Wales. The Hybrid Procedure is currently only offered to babies with HLHS who have a significant extra risk that would affect their survival from the surgical Norwood. The reason for this choice of treatment may be because the baby is very small or premature or they have other abnormalities outside the heart. These added complications increase the risks for these babies. Your cardiac team will explain why each option may be better for your baby.
Longer-term outcomes for children who have followed the whole treatment pathway
There is less information about the much longer-term outcome for babies born with HLHS who undergo a Norwood procedure.
In Australia and New Zealand they collect information about patients who had a Fontan in a Registry and by doing this they have found that about three-quarters of people having a Fontan procedure, the third operation, are still alive 25 years after they had their Fontan operation.
Remember your child is an individual and will have individual risks. Talk to your medical team to understand what they think the risks are for your child. It is important to understand that although children have an improving survival rate following surgery they will still only have half a working heart: this will affect their day-to-day life. Here is a small snapshot of areas of their life that might be affected.
Energy
Children with half a working heart have a reduced amount of energy that they can use every day. This does not usually confine them to a wheelchair or stop them taking part in activities, or even some sports, but it can affect their ability to concentrate and their ability to keep up with their friends.
Feeding
Babies with half a working heart require more energy to run their body and to grow. Sadly, they do not have the energy to take in more food/ milk so often they need help to gain more calories. Support will be offered to new parents as they learn the different techniques required to ensure babies receive the calories they need.
Growth and development
Most babies with half a working heart start their lives at a normal new-born baby size. They have to have a number of operations in their first year and they need a great deal of energy to grow, so they may grow slowly at first. They are very likely to meet all their developmental milestones, as measured by the health visitor, but towards the end of the normal time frame rather than the beginning. For example, some babies walk at nine months, and others not until 18 months. It would be more normal for a baby with half a working heart to walk at the later end of the range.
Schooling
As stated above, children with half a working heart lack energy and so can find the school day difficult. Planning the day, maintaining concentration and completing homework can be a challenge. Development and educational challenges are present in one third of children with a one pump heart, but as in all walks of life, a percentage of the children will do very well in school, while some children will need more educational support.
Other Interventions
Although the plan set out when a child has their first diagnosis usually involves three stages of surgery, many children require other procedures to improve their heart function as they get older. Here we briefly describe some of the possible interventions that may be needed.
Ballooning a blood vessel
Children who have malformations within the heart that require reconstruction of blood vessels, often develop a narrowing around the site of the surgery or the malformation. To ensure that blood can flow freely through the blood vessels, cardiologists often balloon the area of the narrowing to stretch it back to normal or nearly normal size. The ballooning takes place during a cardiac catheterisation. Most children have this procedure done under a general anaesthetic and they may be in hospital for an overnight stay.
Inserting a stent
If widening a vessel with a balloon is not effective or if it is thought that the result of ballooning will not be long lasting, it may be necessary to insert a stent (a mesh of fine wire in a tube shape) that can be used to keep the blood vessel open after it has been stretched. This is inserted during a cardiac catheterisation.
Surgery to a heart valve
Children who rely on one heart pump and not the normal two have more blood flowing through the working side of the heart than normal, until the third stage of surgery has been completed. This added blood flow increases the strain on the valve between the collecting and pumping chambers by stretching the heart valve, creating a leak. If the valve shows signs of long-term damage that will not be relieved by the Fontan operation, surgeons may decide to repair and on rare occasions replace the valve. This involves open heart surgery and a post-operative trip to PICU (Paediatric Intensive Care Unit).
Creating or closing a fenestration
Many children undergoing the Fontan operation will have a hole, a fenestration, created to help blood flow smoothly through the lungs and to reduce the pressure on the circulation. The fenestration creates a hole that allows oxygen-filled red blood and deoxygenated blue blood to mix; mixing the blood means that oxygen saturation levels will not reach 100%. As the children grow it may become clear that the fenestration is no longer open or no longer needed so a plan will be made to re-open or close it with an umbrella device placed in the heart. The procedure is undertaken during a cardiac catheterisation (see Cardiac tests). Most children have this procedure done under a general anaesthetic and they will be in hospital for an overnight stay.
Removing collaterals
If a child is very blue (cyanosed), the body tries to correct the problem by creating extra blood vessels to take blood to the lungs where it can pick up oxygen. These blood vessels are known as collaterals. Often these extra vessels create problems for the child’s Fontan circulation and they need to be closed off. This may happen during planned surgery or during a cardiac catheterisation. For more information on collaterals, visit www.www.lhm.org.uk, Information section.
Heart rhythm
A heartbeat is created by an electrical message that passes through the heart and tells the heart muscle to push blood from one section of the heart to another and then out of the heart to the circulation. The beat (pulse) created depends on the body’s activity, so at rest the rate is lower and on exercise it becomes faster. Children and adults with congenital heart conditions may have a disturbed heart rhythm (arrhythmia). Sometimes they are born with an arrhythmia
and sometimes one develops. Many children who have had major heart surgery have a disturbed heart rhythm after their operations. Arrhythmias are usually
temporary and resolve as the heart recovers from surgery. In some cases the heart rhythm does not recover and a more permanent solution will need to be found. The first treatment offered will often be medications to control the heartbeat. This may solve the problem and no further intervention will be needed. For some children and young adults other solutions will need to be explored.
Ablating an electrical pathway
In some patients the electrical pathway will be disturbed so that heart rhythm messages will be sent to the wrong part of the heart, which creates an imbalance with the pumping of the heart muscle. In these cases cardiologists may offer ablation as a treatment. This is a treatment undertaken under anaesthetic in the cardiac catheterisation theatre. Cardiologists identify the pathways of faulty
electrical activity and block the ones that are creating an imbalance. This may be done by either freezing or burning the faulty pathway.
Pacemaker
If an arrhythmia (abnormal heart rhythm) persists and affects the heart function, doctors may decide to insert a pacemaker. There are a number of different pacemakers that solve different problems. Some help to regulate the heartbeat, others send an impulse to the heart when there are long pauses between beats. Surgery will be needed to insert the pacemaker but it is not as complex as open-heart operations. The pacemaker box is either placed in the abdomen, especially in smaller children, or under the arm or chest muscles in an older/bigger patient.
Following a pacemaker insertion, regular pacemaker checks will be set in place. Changes to the pacemaker settings can be made externally so there is no need for further invasive interventions until the pacemaker box needs changing – usually at around ten-year intervals.
ECMO/ECLS (Extracorporeal Membrane Oxygenation/Extracorporeal Life Support)
Babies and children with congenital heart conditions sometimes have periods of time when they have difficulty pumping enough oxygen-filled blood around their body. This may be because their heart is failing as they await surgery or else occurs in the period after an operation when their heart is tired. If doctors feel that a period of added support for the heart will aid recovery, they may suggest ECMO/ECLS.
The ECMO/ECLS machine is similar to the heart-lung bypass machine used for open-heart surgery, although it is often smaller. Extracorporeal means ‘outside the body’, and a membrane oxygenator is a piece of equipment which delivers oxygen into your child’s blood. Therefore, ECMO/ECLS is the use of an artificial lung (membrane) located outside the body (extracorporeal) that puts oxygen into the blood (oxygenation) and continuously pumps this blood into and around the body.
Most congenital heart units are able to offer ECMO/ECLS for cardiac patients but occasionally a child will be transferred to another cardiac team for this specialised care.
Medications
A very important part of the treatment of complex congenital heart disease is the medical support given to the heart and circulation. This support will be altered as the needs of each child and young adult change as they undergo surgery and grow.
Diuretics
Diuretics are medications that make the kidneys work more efficiently to create urine from the fluid circulating in the blood. The more fluid in the blood, the harder the heart has to work to pump it around the body. It is important to encourage children to continue drinking normally whilst on these medications.
Frusemide (Furosemide) is the most commonly used diuretic; it works well in all age groups.
Amiloride and Spironolactone are often used to keep the body’s salts (electrolytes) in balance as the child passes out more urine. On some occasions these medications are used together.
Medications that strengthen or support the heart’s function
Digoxin is a drug that slows, strengthens and steadies the heart. It is sometimes used in the treatment of heart failure as it makes the heart beat more efficiently. It is also occasionally used to treat heart rhythm problems.
Captopril, Lisinopril, Ramipril and Enalapril – ACE (angiotensin converting enzyme) inhibitors are medications that open up the body’s
blood vessels, making it easier for the heart to pump blood through
them. This relieves pressure/workload on the heart pumps (ventricles).
Carvedilol is a drug that slows down the resting heart rate and
increases the efficiency of the heart muscle.
Anti-arrhythmics
These are medications that help a heart that is not beating in
rhythm. These drugs strengthen the heartbeat and make the beat
more regular.
Digoxin is a drug that slows, strengthens and steadies the heart. It is sometimes used in the treatment of heart failure as it makes the heart beat more efficiently. It is also occasionally used to treat heart rhythm problems.
Amiodarone helps treat heartbeats that are irregular by blocking some defective electrical signals. Avoid eating grapefruit if prescribed this medication because it alters the function of Amiodarone.
Beta blockers, e.g. Sotalol, Propranolol, Bisoprolol are prescribed to block irregular electrical messages as they pass through the heart. This steadies the heartbeat.
Anticoagulants
These are drugs that lengthen the time it takes for the blood to clot. They are used to ensure that the blood flows freely through the tubes and passages that have been created in the heart. For more information, see the LHM booklet Living with anticoagulation.
Aspirin is a gentle anticoagulant that stops blood cells sticking together. It is often the drug of choice in young children.
Warfarin is a medication that slows down the blood’s ability to clot (form scabs). Regular blood tests are required to ensure that the correct dose of Warfarin is given.
Dipyridamole works to interrupt the formation of clots.
Clopidogrel works to interrupt the formation of clots. It sometimes replaces Aspirin or can be used in partnership with Aspirin.
Anti-pulmonary hypertensives
These are specific to the lungs.
Sildenafil works to relax blood vessels so that they widen more easily, making it easier for the blood to flow through them.
Dipyridamole works to reduce blood pressure by opening up blood vessels. It works especially well in the lungs.
Challenges, consequences and complications on the single ventricle treatment pathway
This section explores the sort of challenges that can occur before, during or after heart surgery when a child or young adult has a single ventricle heart.
When doctors discuss a new baby’s diagnosis and the treatment path that they plan for them to travel, they also discuss the risks that go hand in hand with a complex heart problem and the treatments available.
Most parents understand that having only half a heart is never straightforward.
Here we briefly discuss a broad range of problems that can occur. It is very important that parents, and the patients themselves as they get older, talk to their medical team about their personal risks as not all problems are a risk for every child.
Reduced energy levels
When you only have one ventricle doing the job of two it affects the amount of energy you have for day-to-day life. Learning to balance
your activity with the amount of energy available every day is something that children gradually learn to do.
In early life the area most affected by reduced energy is the ability to take in calories and grow.
Feeding
Babies with half a working heart need more calories to run their inefficient heart but their heart condition affects the amount of
energy available to take in any extra calories. To help with their growth, babies will often be offered higher-calorie milk and have a mixture of feeding styles. For example, the baby may breast and bottle feed or bottle and nasogastric tube feed (a small tube passed through the nose into the stomach).
Low oxygen levels
All children with a cyanotic heart condition have lower-than-normal oxygen levels. It is safe for them to be moderately low for short periods of time, especially when there is a treatment plan in place and the children are carefully monitored, but if oxygen levels are left too low for long periods of time, this can affect energy levels and brain function.
There is also a risk of low oxygen levels when a child undergoes cardiac surgery or in the postoperative recovery period, especially if the team uses the heart-lung bypass machine. Low oxygen levels leading to problems with the brain are known as neurological incidents.
Doctors will explain the risk of low oxygen levels but they will also work to reduce that risk as much as they can.
Infection
Postoperatively there is a risk that the chest scar wound both inside the body and on the skin will become infected. The doctors work to prevent this by giving antibiotics but it can still occur after surgery. If the scar is weeping or red, a test will be done to identify the type of infection and treatment will begin.
Necrotising Enterocolitis (NEC)
NEC occurs when part of the gut (intestines) starts to die and becomes infected because of a lack of oxygen. This can occur in babies around the time of the first stages of surgery, causing an intolerance to the absorption of milk in the gut. Treatment will be given for the infections and the gut will be rested; calories will be given via a drip (intravenous line). Babies may require surgery to remove any of the gut that has died.
Bacterial endocarditis
There is also a risk of something called bacterial endocarditis. This is an infection that often starts in the mouth and can travel to the heart. For more information on dental care, visit the information section of our website.
Blood loss
After any operation there is a risk of bleeding. Blood loss will be measured and replacements will be given if the medical staff feel that the loss is compromising blood flow. If the blood pressure remains low for long periods, the kidneys are unable to work properly and ultimately blood containing oxygen is unable to reach the brain.
If bleeding occurs between the heart muscle and the outside skin of the heart, this can cause problems with the pumping of the heart. If this happens, the surgeons may need to remove the blood that has collected there.
Chest drain losses
After chest surgery children and adults have chest drains inserted to ensure that any fluid or blood in the chest can drain away. In some cases the drainage becomes excessive. This could signify increased blood loss or that fluid fat (chyle) is leaking in the chest. If the drainage is excessive, and doctors are unable to keep the fluid in balance, they may require a change in diet or further surgery to look for a bleeding or leak point.
If chyle continues to collect in the chest, the children will be prescribed a change in their diet. The MCT (medium-chain triglyceride) diet reduces the amount of fat a child eats. This helps to reduce the amount of chyle in the chest, allowing for any leaking points to heal. For more information, see the LHM booklet MCT diet.
High blood pressure in the lungs
The need to change the way that blood flows through the heart and around the body can create changes to the blood’s pressure in different parts of the body.
Normally there is a low blood pressure in the lungs but following the second and third stages of surgery, or as the child grows from childhood to adulthood, the pressure can change.
If the blood pressure rises in the lungs, it creates a back-pressure on the liver, gut and other parts of the body normally below the lungs.
Without treatment this can create changes to the way that the liver and gut work. For more information, see the long-term effects of the Fontan within The Fontan procedure booklet.
Clots
The heart is responsible for pumping blood around the body. If the heart is not working efficiently, especially around the time of surgery,
there is a risk that the blood will clot on areas where surgery has been performed or in pockets of the heart where blood moves slowly. There are also changes to liver function in patients who have a Fontan circulation, and these make the risk of clotting greater than normal. There are a number of risks associated with this clotting. The heart’s pumping can be compromised if clots grow within the heart. Small bits of the clot can travel into the lungs, causing problems with breathing, or to the brain where they could cause a stroke. Children and young adults undergoing heart surgery or living with a Fontan circulation are often prescribed anticoagulation medication to reduce the risk of clots. See LHM’s booklet Living with anticoagulation.
Strokes
A stroke is a disruption to the circulation of blood to the brain, either because of a bleed or because of a blockage, usually a clot. This disruption causes damage to the brain tissue, a condition that is called a stroke. Strokes can take different forms. They can affect mobility, sight, speech and memory, and can cause developmental. problems. Medical teams will be working to avoid strokes by making sure that the risk of clots is low, and that the heart is supported to pump well and maintain the oxygen circulating to the brain.
Arrhythmias
An arrhythmia is a problem with the rate or rhythm of the heartbeat. During an arrhythmia, the heart can beat too fast, too slowly, or with an irregular rhythm. Children with single ventricle hearts have a risk of arrhythmias because of the structure of their heart. The risk is increased after surgery when the circulation has been altered. Arrhythmias can be treated with medications or the use of a pacemaker but often a postoperative arrhythmia will disappear once the heart has recovered from the operation.
A number of these complications can occur routinely around the time of surgery or treatment and so the medical and nursing staff will be watching for them and will intervene swiftly with treatment to help resolve the problems
Identifying key hospital and community staff
When you first enter the world of congenital cardiac care it can be very difficult to know who everyone is and what they do.
In this section we hope to help you understand a little more about the people you might meet, from the time a baby is diagnosed with a complex heart condition and onwards.
Antenatal team
The antenatal team is usually made up of a number of obstetric (pregnancy) and cardiac staff.
Midwife
The nurse specially trained to help with the pregnancy and delivery of the baby.
Obstetrician
The pregnancy and delivery doctor.
Sonographer
The member of staff who performs the ultrasound scan on mothers to look at the baby.
Fetal obstetrician
A doctor who specialises in looking at the unborn baby, especially if there are problems with the way that the baby is developing. They will look at all parts of the baby and offer other tests if they detect any problems before the birth.
Fetal cardiologist
A cardiac doctor who specialises in looking at heart problems in an unborn baby. They may also be a children’s cardiologist who looks after children with a heart condition. They will explain any problems with the baby’s heart and set out the treatment plan to treat any condition.
Fetal midwife
A nurse who will help parents understand an antenatal diagnosis, plan tests and link families with local and specialist maternity services.
Congenital cardiac specialist nurse
A nurse who specialises in supporting and informing families about the cardiac diagnosis and treatments. They provide a link from the obstetric team to the cardiac team.
Congenital cardiac team
Congenital cardiologist
The doctor who makes a diagnosis of congenital heart conditions and then looks after the child or adult patient throughout their life. They organise tests and see patients both in the hospital and at outpatient appointments. They also prescribe medications. Some cardiologists do interventional cardiac procedures (not surgery) during cardiac catheterisation, for example, closing small holes in the heart.
Congenital cardiac surgeon
A doctor who offers surgical solutions to children and adults who have a congenital heart condition. They manage the care of a child or adult if they are admitted to hospital for an operation. 54
Congenital cardiac specialist nurse
A nurse who has extra training and experience in looking after children and/or adults with congenital heart conditions.
Congenital cardiac ward nurse
A cardiac nurse who looks after a child or adult who has been admitted to the hospital to undergo tests or treatment for a congenital cardiac condition.
Anaesthetist
A doctor who is responsible for keeping a child or adult asleep when they undergo certain tests or surgery.
Intensivist
A specialist doctor who treats and observes a child or adult whilst they are in the Paediatric Intensive Care Unit (PICU).
PICU/ITU (Paediatric Intensive Care/Intensive Therapy Unit) nurse
A nurse with special training to look after a very sick child or adult in the intensive care ward.
HDU (High Dependency Unit) nurse
A nurse with special training to look after a child or adult who needs high dependency care. This is a step of care between intensive care and general care and usually takes place on a high dependency ward or a specially equipped area of a cardiac ward.
Cardiac physiologist and/or sonographer
Specially trained staff who work with cardiologists to undertake medical tests such as echos (heart scans) or ECGs (recordings of the heart rhythm).
Psychologist
Specially trained staff who can help with the stress and strain that can be created when a child has a complex heart condition, for example, fear of being in the hospital or fear of needles.
Dietitian
An expert in knowing what food and drink is needed to keep healthy. They provide very important support for families learning how to help their children grow and thrive. Some children are referred to a Speech and Language specialist for added help with feeding because they specialise in how the mouth and throat work.
Hospital social worker
A member of staff trained to support parents, families and patients with the social care they may need when they leave hospital. They can help with allowances and benefits.
Play specialist
Helps children recover from operations or prepare for treatment by encouraging activity and fun play. They are based in the ward or play department
Understanding common terms
The following are terms that you may hear whilst your child is in hospital or at home receiving treatment. There will always be some terms that are not covered, so ask your cardiac team to explain things if they use terms that you do not understand.
Hypoplastic
Small or underdeveloped
Atrium
Collecting chamber of the heart
Ventricle
Pumping chamber of the heart
IVC (Inferior vena cava)
Lower body vein
SVC (Superior vena cava)
Upper body vein
Stenosed
Narrowed
Atretic
Not fully formed/closed
Patent
Open
Cyanosed/cyanotic
Blue colour due to lack of oxygen
Acyanotic
Not looking blue
Oedema
Fluid retention, puffiness
Pyrexia
High temperature
Apyrexia
Normal temperature
ECG (electrocardiogram)
A recording of the electrical activity of the heart
Echo (echocardiogram)
Uses sound waves to scan the heart which is then shown as a picture
ASD (Atrial Septal Defect)
Hole between the top two collecting chambers of the heart
VSD (Ventricular Septal Defect)
Hole between the bottom two pumping chambers of the heart
AVSD (Atrioventricular Septal Defect)
A large hole in the centre of the heart affecting the wall between both the collecting and pumping heart chambers
We would like to thank members of our reader panel for reviewing this information and sharing their experiences.
References are available from the LHM office on request.

